Proofreading Services
Professional editing and proofreading services.
Submit your work with confidence, knowing it’s free of language errors and flows well from beginning to end.
- Ensure your arguments are judged on merit
- Lift the quality of your paper as a whole
- Make you stand out from your peers

- Proofreading & Editing

Native editors
Become a better writer, 100% happiness guarantee, pick your proofreading and editing service.
Stand out from your peers with an error-free thesis that flows well from beginning to end.
- PhD dissertations
Impress your supervisor and graduate with a flawless dissertation.
» Dissertation editing
Earn higher grades on your essays with the essay checker. » Essay Checker
Research & term papers
Boost your grade with a well-structured, clear, and error-free paper.
» Paper editing
Manuscripts & academic articles
Improve your chances of publication with a polished manuscript.
» Academic editing
- Capstone projects
Demonstrate your expertise and obtain your degree with a perfect capstone project.
Secure funding or approval from supervisors with a well-written proposal.
English documents
Sound like a native English speaker and communicate your thoughts clearly.
» English proofreading
Literature reviews
Set the foundation for a great piece of academic writing with a well-written literature review.
Professional documents
Boost your career with an outstanding resume, personal statement, or cover letter. » Professional proofreading
Business documents
Ensure that your business documents, including reports, presentations, and web content, are professional in tone. » Business proofreading
Online Proofreader
Write your paper, thesis, or dissertation stress-free. Fix all your grammar, spelling, and punctuation mistakes instantly with 1 click.
» Online proofreader
Free AI Writing Resources
Improve your academic performance and write like a pro. Get access to free AI writing tools, useful ChatGPT prompts, the latest articles about AI, and more.
» Free AI writing resources
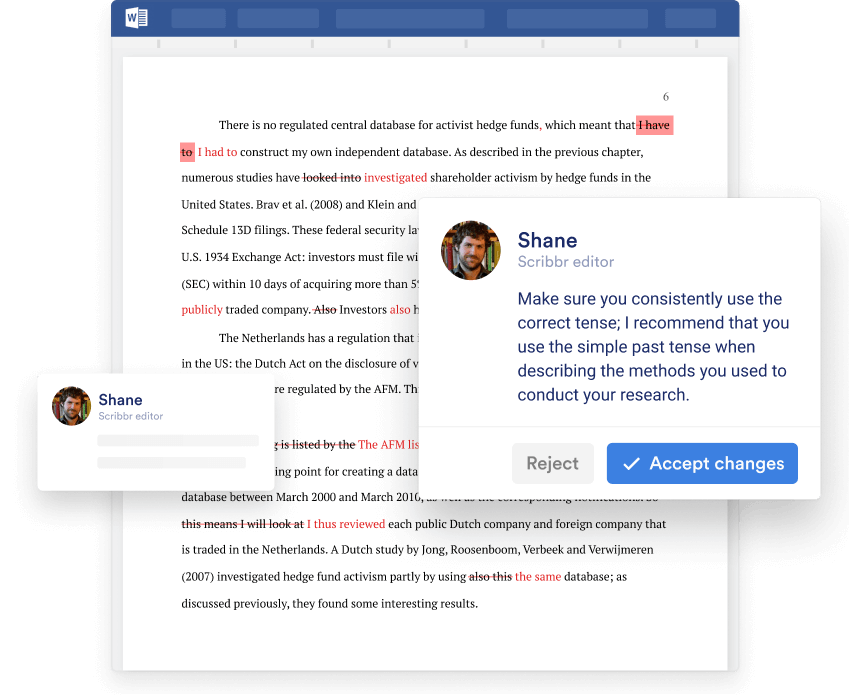
The Scribbr editing service
This is how we improve your document.
Standard Proofreading & Editing is perfect if you’re confident about your writing but need a second pair of eyes to catch:
- Spelling and grammar errors
- Inconsistencies in dialect
- Overuse of passive voice
- Subjective or inflated language
For a more comprehensive edit, you can add one or multiple add-on editing services that fit your needs.
| ⏰ Deadline | Within 3 hours |
|---|---|
| 📄 Texts | Papers, essays, reports, manuscripts |
| ⭐️ Rating | based on 13,917 reviews |
Add-on services
Customize your editing package to get the help you need, structure check, clarity check, paper formatting, citation editing.
Ensures sections and chapters are structured and focused and your writing is free of redundancies.
- Through in-text feedback, your editor will help:
- Organize and focus individual chapters and sections
- Eliminate repetitive and redundant information
- Perfect transitions between sentences and paragraphs
- Align titles and headings with the section’s content
You’ll also receive a personalized Structure Check Report meant to help you identify missing elements in each chapter or section and prioritize improvements.
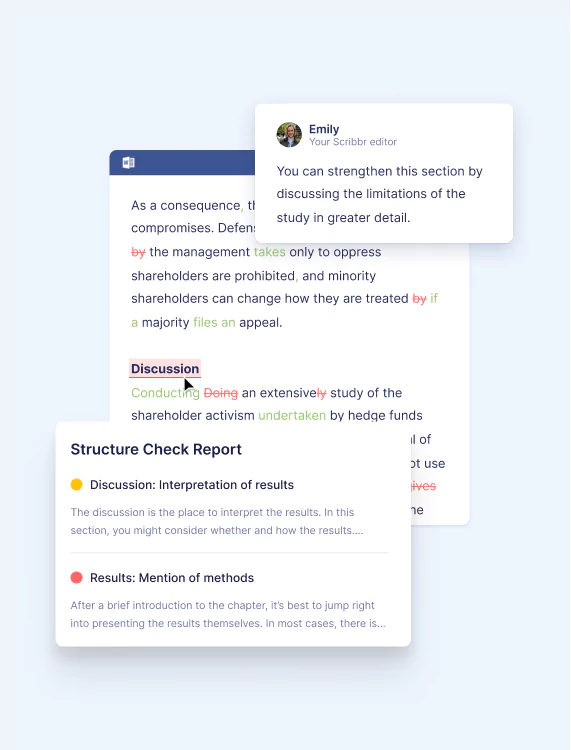
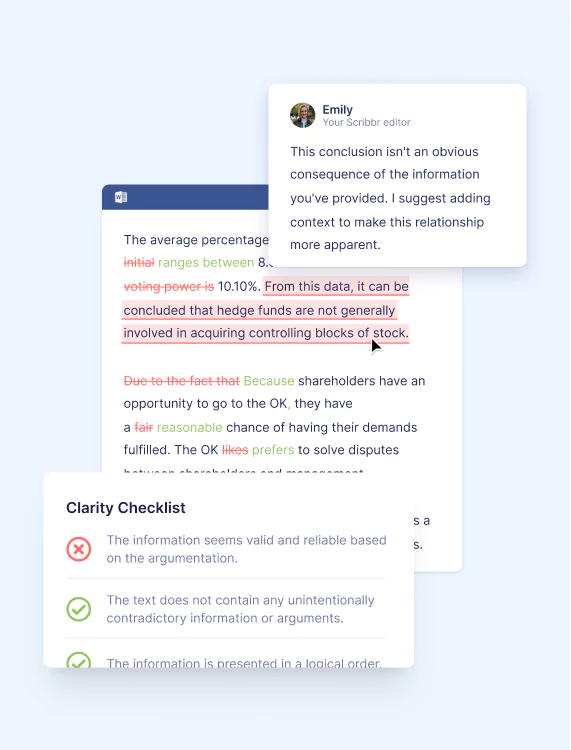
Ensures ideas are presented clearly, your arguments are consistent, and your audience can follow along.
Through in-text comments and checklists, your editor will:
- Make sure your text tells a clear and logical story
- Check that you’ve clearly presented concepts, ideas, and key terms
- Make sure your key takeaways and conclusions are front and center
- Highlight contradictions within the text
- Ensure you’re keeping your audience’s needs in mind
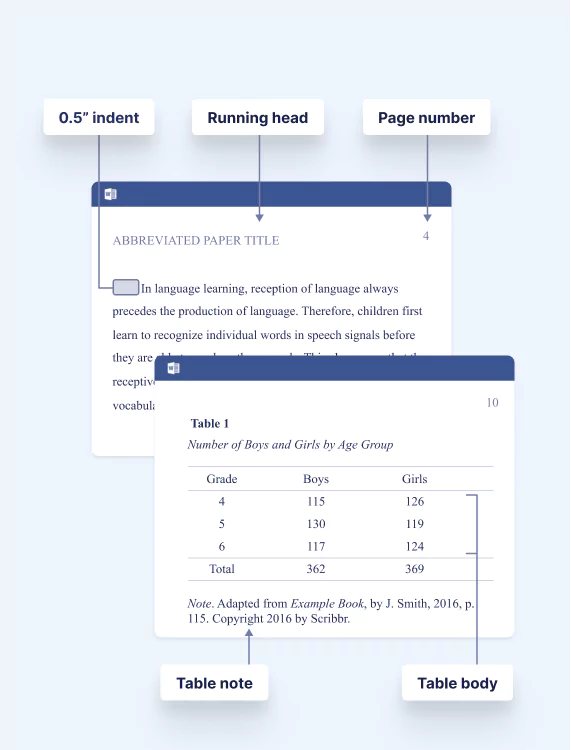
Ensures a professional look for your document that meets your formatting requirements.
Your formatting expert will ensure consistency for the following:
- Margins, spacing, and indentation
- Body text and headings
- Page numbers
- Abstract and keywords
- Explanatory footnotes
Choose our Paper Formatting service for a professional finish or our APA Editing Service for the most up-to-date APA formatting.
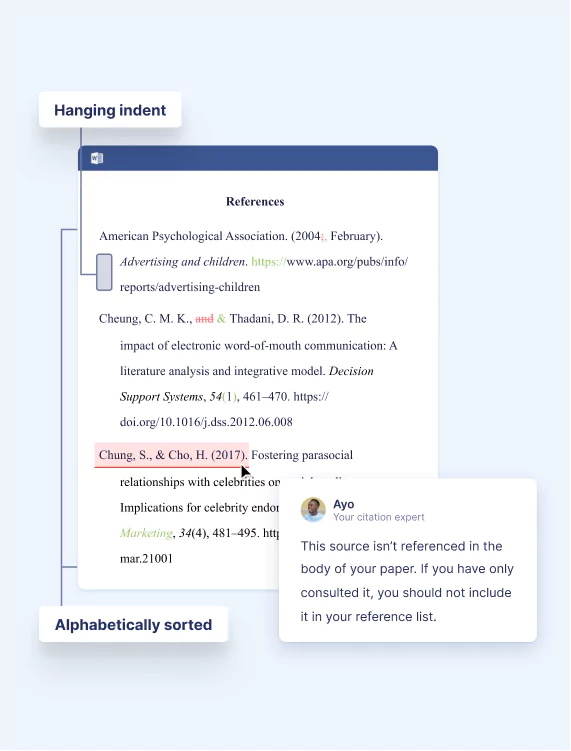
Citation Editing ensures your citations and references are consistent and meet your style guide’s requirements.
After you provide your document with a reference list, your citation expert will:
- Format the layout of your reference page (margins, indents, spacing)
- Ensure that your chosen citation style is applied consistently according to the guidelines
- Cross-check citations with reference entries
- Provide feedback on reference list entries that you need to complete due to missing information
Your expert is familiar with all common citation styles. Find more information about the service and our requirements in our FAQs .
Native academic editors
You'll only get matched with best editors.
At Scribbr, you can rest assured that only the best editors will work on your paper.
All our 800+ editors have passed the challenging Scribbr Academy, which has a passing rate of only 2%. To put that in perspective, Harvard has an acceptance rate of 3%.
We handpick your editor on several criteria, including field of study and document type. And we’ll even expand your team with citation and formatting experts if needed.

I have a doctorate in biology and studied a range of life science subjects. I specialize in editing academic texts.

I researched at Harvard, taught English with a Fulbright in Peru, and earned a master's from Johns Hopkins.

I am an academic editor and book reviewer. I am familiar with many style guides and have edited over 6 million words.

I have a bachelor's in electrical engineering and a master's in psychology and am pursuing a PhD in neuroscience.

I am an ESL teacher and academic editor with a research background in the humanities, arts, and culture.
Improve your writing with professional proofreading and editing services
Same day delivery
This deadline works automatically for the following document sizes:
- 3 hours: less than 3,000 words
- 6 hours: less than 6,000 words
- 12 hours: less than 12,000 words
Select your currency
“Always feel empowered after the proofreading”
This is my third time using Scribbr. I find the proofreading and comments very helpful and caring. I always feel empowered after the corrections.
How it works
This is what you can expect from our proofreading service, upload any time.
Upload your document and easily select the pages that need editing. Next, choose your turnaround time and services, and explain your situation and needs to the editor.
Stay in the loop
After placing your order you can keep track of our progress. From finding your perfect editor to potential hand-overs to formatting or citation experts.
Revise and submit
You’ll receive your document back with tracked changes and feedback, as well as a personal letter from your editor. The last step is submitting your work with confidence!
Scribbr & academic integrity
Scribbr is committed to protecting academic integrity. Our proofreading service, our AI writing tools ( plagiarism checker , paraphrasing tool , grammar checker , summarizer, Citation Generator ) as well as our free Knowledge Base content are designed to help students produce quality academic papers.
We make every effort to prevent our software from being used for fraudulent or manipulative purposes.
Your questions, answered.
Scribbr specializes in editing study-related documents . We proofread:
- Research proposals
- Personal statements
- Admission essays
- Motivation letters
- Reflection papers
- Journal articles
The fastest turnaround time is 12 hours.
You can upload your document at any time and choose between four deadlines:
At Scribbr, we promise to make every customer 100% happy with the service we offer. Our philosophy: Your complaint is always justified – no denial, no doubts.
Our customer support team is here to find the solution that helps you the most, whether that’s a free new edit or a refund for the service.
Yes, if your document is longer than 20,000 words, you will get a sample of approximately 2,000 words. This sample edit gives you a first impression of the editor’s editing style and a chance to ask questions and give feedback.
How does the sample edit work?
You will receive the sample edit within 12 hours after placing your order. You then have 24 hours to let us know if you’re happy with the sample or if there’s something you would like the editor to do differently.
Read more about how the sample edit works
Yes, in the order process you can indicate your preference for American, British, or Australian English .
If you don’t choose one, your editor will follow the style of English you currently use. If your editor has any questions about this, we will contact you.
Yes, regardless of the deadline you choose, our editors can proofread your document during weekends and holidays.
Example: If you select the 12-hour service on Saturday, you will receive your edited document back within 12 hours on Sunday.
Our APA experts default to APA 7 for editing and formatting. For the Citation Editing Service you are able to choose between APA 6 and 7.
Every Scribbr order comes with our award-winning Proofreading & Editing service , which combines two important stages of the revision process.
For a more comprehensive edit, you can add a Structure Check or Clarity Check to your order. With these building blocks, you can customize the kind of feedback you receive.
You might be familiar with a different set of editing terms. To help you understand what you can expect at Scribbr, we created this table:
| Types of editing | Available at Scribbr? |
|---|---|
| | This is the “proofreading” in Scribbr’s standard service. It can only be selected in combination with editing. |
| | This is the “editing” in Scribbr’s standard service. It can only be selected in combination with proofreading. |
| | Select the Structure Check and Clarity Check to receive a comprehensive edit equivalent to a line edit. |
| | This kind of editing involves heavy rewriting and restructuring. Our editors cannot help with this. |
View an example
Your editor is on stand-by and ready to start editing your document.
Get in touch, with real people.
We answer your questions quickly and personally from 9:00 to 23:00 CET

- Start live chat
- Email [email protected]
- Call +1 (510) 822-8066
- WhatsApp +31 20 261 6040
Knowledge Base
Level up your writing with scribbr’s top-rated guides.
Academic Writing
Language Rules to Improve Your Academic Writing
The dos and don’ts for academic writing, english mistakes commonly made, common word choice confusions in academic writing, ask our team.
Want to contact us directly? No problem. We are always here for you.
Frequently asked questions

Scientific Editing
Brought to you by Springer Nature
Get expert in-depth editing and strategic advice on your manuscript, review article or grant proposal from a team of editors following the quality standards set by Nature Research. Scientific Editing includes a Gold English language edit, a developmental edit, a quality assurance edit and a summary report. We specialize in research papers, reviews and grant proposals within the natural sciences (medicine, biology, chemistry, physics and earth sciences) and engineering.
A Team-based Assessment of Your Manuscript’s Needs
Scientific Editing team members meet to identify priority issues in the manuscript and discuss how the priorities can be addressed. We leave comments about additional documents provided for editing, such as cover letters or responses to reviewers. We provide insights into how readers, peer reviewers, and journal editors might view your paper.
A Thorough Developmental Edit
A Substantive In-Depth Assessment of Your Scientific Manuscript
Based on the priorities identified in the team meeting, experienced members of the Scientific Editing team examine the text in significant detail and provide the following:
- Strategic developmental edits that will improve the structure and clarity of the logical argumentation, such as how well the implications of the work are highlighted
- Incisive comments recommending further author revisions that will help you maximize your manuscript’s effectiveness.
- Improved English grammar to ensure that your ideas are conveyed clearly
A Thorough Strategic Report
Customized to the Needs of Each Scientific Manuscript
Scientific Editing also includes a custom 3-6 page report that includes:
- Explanations of the prioritized issues identified in your paper, how we addressed them and/or how you might be able to address them further.
- Insights into how readers, peer reviewers, and journal editors might view your paper
- Strategic advice for improving your writing in the future.
- Comments about additional documents provided for editing, such as cover letters or responses to reviewers.
Continued Support after Scientific Editing
Trusted by Researchers and Universities to Help their Science Stand Out
- After you have received Scientific Editing and have considered the advice and made changes, we provide one free language edit (for field-specific grammar, phrasing, and punctuation correction) within 6 months of the completion of the initial order.
- You may also want to purchase Manuscript Formatting at this time; from our partners, AJE

Download one of our sample files to see the types of suggestions and edits made by our Scientific Editors
Confidentiality Assured
We never show your paper to anyone so you can be confident that when you submit to a journal, the editor’s decision will be independent and fair
These policies relate specifically to Nature Research Editing Service. Please see also our Terms of Service for other core principles, for example regarding the confidentiality of your documents. Our aim is to serve the interests of authors and institutions, not the editorial and publishing interests of Springer Nature journals. We openly and clearly declare to potential and current customers that we are part of Springer Nature, and we inform potential customers about our independence from the publishing process. We aim to be publisher-neutral in our editing and advice, and especially in our journal recommendations (where applicable). We take steps during the editing process, when giving advice, and when recruiting and training editors to help us achieve this aim. Nature Research Editing Service does not measure its success in terms of the publication of edited documents by Springer Nature journals. We do not involve ourselves in the publishing process. We do not guarantee publication of papers or funding of grant applications. We expect journal editors and grant review panels will make these decisions entirely independently of Nature Research Editing Service and other author services providers. Draft journal articles and their scientific content are not shared with professional editors who make decisions on research papers at Springer Nature journals. Any comments that we make regarding the suitability of specific target journals are explicitly presented as opinion and not as a definitive prediction of the outcome of submission. If an author indicates that they mainly want to know whether or not their paper is acceptable to a specific Springer Nature journal, or has any other query for which a Springer Nature journal editor is the most appropriate contact, we will suggest that the author contacts the journal directly. We do not suggest peer reviewers. We do not infringe on authorship. When editing documents, wherever possible, we provide our edits and comments in an easily reviewable format. We encourage authors to review our input critically, rather than accepting all of our edits and suggestions without review. We never confirm that any document is ready for submission, submit anything to a journal or funding body, or correspond with journal editors or peer reviewers on behalf of authors. We do not ghostwrite or completely rewrite large passages of text, and we do not directly introduce ideas that are not already present in the documents submitted to us. Our editing focuses on improving how the existing research is communicated. General policies We do not engage in or encourage practices that contravene relevant ethical guidelines. For Scientific Editing, if we suspect unethical practice in a submitted document or correspondence, we will point it out and explain it. If we suspect severe infringement of ethical requirements or laws (particularly unacceptable harm to humans or animals), we act on this where possible. We review these policies on a regular basis and keep up-to-date with issues arising in the publication ethics landscape. We train editors to follow these policies, we monitor adherence to these policies, and we take action in the event of any failure to follow them.
Our Quality Guaranteed
To ensure that the changes you make based on the editor’s report are grammatically correct, we include one free language re-edit of your revised paper before you submit to the journal (for Scientific Editing only). To claim this free re-edit, email support within 6 months of your original order and attach your revised paper. After this free re-edit, we guarantee that if your language-edited paper is rejected by a journal because of the English alone, we will re-edit your paper once for free.
Where Our Customers Publish
- Advanced Functional Materials
- Cancer Research
- Developmental Cell
- Earth and Planetary Science Letters
- Energy and Environmental Science
- Genome Biology
- Geophysical Research Letters
- Journal of Experimental Medicine
- Nano Letters
- Nature Climate Change
- Nature Communications
- Nature Genetics
- Nature Geoscience
- Nature Medicine
- Proceedings of the National Academy of Sciences of the United States
- Science Advances
Testimonials
I would like to express my sincere appreciation for your work. You did really an excellent job. What I liked in particular was that you gave comments with substantial suggestions how to improve. Although I have published till now quite a number of scientific papers (>300) and many of them in high impact journals I learned a lot throughout this. I Iook forward to future cooperation of mutual benefit. -Full Professor · University of Bayreuth · Germany
The report with the feedback summarizing edits and comments is very informative. We appreciate a lot your editor’s work, with changes tracked and comments on the text improving clearly our manuscript. Finally, your recommendations concerning the most appropriate journal for publication will decidedly help us for the next submission of our manuscript. -Research Director, CNRS/Team Leader · Sorbonne University · France
- [email protected]
- No.6/387, First Floor, Mogappair East, Chennai.
- GET A QUOTE

Service Beyond Hopes!
Scientific writing services, what does scientific writing services do.
A professional and competent scientific writing services helps you to craft, draft, build and produce an error free scientific article for publication in peer-reviewed high impact scientific journals. We offer the service through a team of experienced and hi-skilled scientific writers. You can use their skills and expertise to build your publication quickly, easily, and effectively.
Why is scientific writing different from other writings?
Scientific writing is different from other forms of writing as it demands extensive experience, maturity, and accuracy in writing. That’s why many research scientists find it difficult to write an accurate research paper. Some even struggle to find the right research topic. So, what do they do? Sit hand in hand, wait for some magic to happen and write their research document? NO! They hire a professional scientific writers.

Who are scientific writers?
The scientific writers are Certified in English for Research Publication Purposes , with a Master’s or doctorate degree in Life Sciences. They help to:
- Improve the style, format, and language of your scientific research paper
- Make the research paper meet the desired journal requirements
- Revise your research paper so that it instantly connects with the journal editors and other readers
- Reduce the time for publication by avoiding multiple drafts
- Increase your chances of publication in the desired journal
If you are hunting for the right, secured, and powerful scientific writing paper services, your search ends at CogniBrain!
Why CogniBrain?
CogniBrain is the hub of proficient scientific writing services company with several decades of experience. With us, you would sense the change that modifies your raw data into a splendid paper ready for publication.
- We accept half-written or rough drafts of the scientific article and turn them into a well-written research document.
- We allow you to focus on your research work while we write the scientific paper for you.
- All our scientific writers are well-versed with the latest trends in health domain.
- We maintain the confidentiality of all your submitted documents.
- We offer cost-effective services within a given time-frame.
- We provide an expert opinion and genuine insights about your research paper from a reader’s point of view.
- We offer scientific editing service to make your paper free from all jargon.
- We recommend valuable and interesting research topics for your thesis or PhD.
Types of Scientific Writing Services Offered by CogniBrain
Original article writing.
An original article pens down the findings of an original research study. Authors conduct a research on a particular topic through experiments, surveys and observations, and report their findings through original scientific research articles.
If you are nervous to draft your original research article or the lab work leaves no time to write your scientific paper, get help from our skilled and experienced scientific writers.
Review article writing
Writing a review article requires keen observation and evaluation skill to research and summarize the published studies on a particular topic, rather than reporting the new facts. Plus, the articles must not be a copy of the previous studies.
Our fully trained review article writers possess the necessary skill and acumen in all the writing patterns to deliver a plagiarism free content in the desired format.
Case report writing
A medical practitioner comes across several complicated cases in his lifetime, where it isn’t easy to find the right treatment. Reporting the facts you learn from such cases can be a boon to your reputation and a great contribution to the Medical Science.
Leverage the skills and expertise of our scientific writers to write a case report of your successful treatment and become a hero amongst your colleagues.
Meta Analysis
Meta analysis is the statistical analysis of the data obtained from multiple studies on a specific topic and summarizes them to give the most precise result. Thus, to write a Meta analysis, you must be an expert in writing as well as in statistics to analyze the results accurately.
However, do not burn the night oil to write your Meta analysis. Hire our expert writers who provide complete support to write a perfectly polished and plagiarism-free Meta analysis.
Systematic Review
Writing a systematic review is time-consuming as it requires an extensive research about the literature on which you will base your research question. You must gain full comprehension of the information you are going to use to create your scientific review.
Our professional writers are always at your assistance to narrow down the research question and make it manageable for further research and systematic review.
Proposal writing
The purpose of writing and submitting a research proposal before starting with the project is to prove that the problems you will investigate are important and what will be the significance of its results to the specific field.
Thus, your research proposal must give an apt description of your research aim and convince all the stakeholders about the project. Since this is not always easy, we offer round the clock support through our proficient writers to write a research proposal that no one can reject.
Short communication
Short communications are short papers written in a concise format to report significant improvements in existing research. The findings or improvements must be reported quickly as the need to disseminate the information is high.
Hurriedness to write short communications can compromise its quality. Therefore, our scientific writers devote every second to write an effective short communication that is delivered in a timely manner and serves its purpose.
Newsletters
Newsletters are the most economical way to announce the upcoming events and updates about an organization to a wide range of people. Thus, they must be informative for the readers and not contain much promotional content.
Writing an effective newsletter has become easy with the team of our talented writers who can write fresh and plagiarism free content and makes it serve its purpose for the organization.
Our Specialty Scientific Writing Domains
Medical domain, surgical domain, dental domain, pharmaceutical, nursing domain, biomedical & life-science domain, physiotherapy domain, alternative therapy domain.

Science Editing Services by SciTechEdit:
Communicate your science effectively.
SciTechEdit provides expert science editing services for all science communications, including scientific research papers, grant proposals, medical communication strategies, and technical manuals.
Our professional science editors work closely with you to produce clear, well-constructed documents describing your scientific, medical, and technical research findings to promote publication in English-language journals, successfully obtain grant funding, and develop compelling scientific communication platforms.
Contact us today to see how we can help you with your science communication needs.

Science Editing
We have a service to meet your needs. learn more, grant proposals, learn how to improve your funding score, materials we edit, learn more about what we edit, try us for free, learn more about our free editing service.
Why is SciTechEdit the best choice for you?
Personalized
Our services are personalized and tailored to your individual needs
Trusted by top scientists around the world
Over 96% of the manuscripts edited by SciTechEdit are published
Professional editors with expertise in a wide variety of fields
SciTechEdit sets the standards for the science editing community
Trustworthy
Our satisfied clients refer us to their colleagues
We have been helping scientists since 1997
Professional, accurate, confidential, and on time
We partner with you to ensure your success
Explore how ScitTechEdit can help with all forms of science communication
Writing Tips
Articles to help you streamline and improve your scientific writing

The Oxford Comma
Learn the importance of the Oxford comma for your scientific manuscripts.

18 Tips to Improve Your Science Writing
18 tips to improve your science writing – organize your thoughts, ideas, and actions in a logical manner – simplify word choices – write concisely.

10 Tips to Streamline Your Writing
How to streamline and improve your science writing process
Our services are personalized and tailored to your individual needs.
Looking for Affordable Editing?
- Discover our competitive pricing and elevate your research. Visit our pricing page now!
Clear writing enhances the reader’s understanding of the importance of your reported findings. Once published, a permanent record of your ideas and hypotheses and thought processes is established, allowing others to build on your ideas for future investigation, which is the backbone of scientific exploration. This is crucial for scientific progress.
Providing other scientists a piece of new information as a platform for building their own inquiries is also crucial for your career as a scientist. Science aims to enhance the understanding and lives of humanity. Findings that cannot be easily accessed or understood delay the advancement of science. The investment, in both time and money, spent on research warrant dispersion of the findings of the research, whatever they may be, to a global audience. The language used must be understood by both native and non-native English-speaking scientists throughout the world. In other words, a more ‘scientific English’ rather than the type of English used socially or in classical literature. Let SciTechEdit help you to communicate your science to an international audience.
Welcome to BMJ Author Services
- 24/7 Service With A Reply In Minutes
- 100% Satisfaction / Your Money Back
“The experience was very good and the premium editing was very helpful. Thank you!”
- Joanna Kowalska
BMJ partners with Editage to offer Author Services to Medical Researchers worldwide and assist them in their publication goals. Editage offers professional editing by specialized subject experts in Medicine . With a comprehensive quality guarantee, Editage promises to assist you until you are ready to submit.
Services we offer
- English Editing Services
- Translation Services
We offer four levels of manuscript editing services. Each service level differs in terms of the scope of edit and post-submission support. All four levels of editing include free manuscript formatting for the BMJ journal of your choice. This will save your time and budget.
| Plus Plan | ||||
| Publication-focused editing that makes your manuscript ready for initial submission. | ||||
| Select this service if you like the editor to help with extensive language revisions, organization of your article, and feedback on your writing. --> | Features | -->|||
| Features | ||||
|---|---|---|---|---|
| Features | ||||
| Revisions for grammar, terminology, word choice, & native English tone | ||||
| Revisions for grammar, terminology, word choice, & native English tone | ||||
| Editing certificate (upon request) | ||||
| Editing certificate (upon request) | ||||
| Letter From The Editor | ||||
| Letter From The Editor | Included in Scientific Review Report | |||
| Unlimited Q&A with editor | ||||
| Unlimited Q&A with editor | ||||
| Intensive edit for organization (rephrasing and restructuring) | ||||
| Intensive edit for organization (rephrasing and restructuring) | ||||
| Customized cover letter | ||||
| Customized cover letter | ||||
| Feedback on original writing | ||||
| Feedback on original writing | ||||
| Unlimited re-edits for 365 days | ||||
| Unlimited re-edits for 365 days | 60% discount | Free | Free | |
| Word count reduction | ||||
| Word count reduction | Word reduction by 10% | Word reduction by 20% | Unrestricted | |
| Journal-specific formatting | ||||
| Journal-specific formatting | Free for one journal | Free for one journal | Free for multiple journals | |
| Journal response letter editing & cross-checking with the revised manuscript | ||||
| Journal response letter editing & cross-checking with the revised manuscript | ||||
| Scientific Review Report which includes: | ||||
| Scientific Review Report which includes: | ||||
| In-depth technical review by top-tier journal peer reviewer | ||||
| In-depth technical review by top-tier journal peer reviewer | ||||
| High-quality developmental editing focusing on paper structure and research design | ||||
| High-quality developmental editing focusing on paper structure and research design | ||||
| Journal submission readiness check by Managing Editor or senior editor | ||||
| Journal submission readiness check by Managing Editor or senior editor | ||||
| Free plagiarism check report | ||||
| Free plagiarism check report | ||||
| | ||||
Don’t get lost in translation! Our academic translators and editors offer subject-specific expertise and superior editing skills to ensure that your manuscript accurately conveys every nuance of your research in perfect scientific English to a global audience. Our aim is simple: deliver a manuscript that journal editors, reviewers and readers find indistinguishable from those written by native English speakers.
We offer subject-specific translation with English Editing for the following language pairs.
- Traditional and Simplified Chinese to English
- Spanish to English
- Korean to English
- Japanese to English
- Brazilian Portuguese to English
- Turkish to English

Why choose BMJ Author Services?
Our goal is 100% client satisfaction. That is what we aim for in everything we do.
From the quality of our editing and translations to the timely delivery of your completed manuscript, we make sure you are satisfied with our service every step of the way.
You reach out to us for expert author services, and we mean to deliver. Our team will work with you until your expectations are met.

- Quality guarantee
- Delivery guarantee
- Confidentiality
- Multiple payment options
Two-Editor Check
Each document is checked by two language specialists from our global team of editors and peer reviewers to ensure that we miss nothing. The end result will meet the highest quality standards of international journals.
RESPONSIVENESS
24-Hour Response Time
Whether you are sending in a submission or query, our customer support managers will respond to you within 24 hours, with solutions customized to your needs. If your document submission is even a second late, we offer a full fee refund, No questions asked!
CONFIDENTIALITY
128-bit SSL encryption
Our ISO/IEC 27001:2013 certified IT security management systems and processes ensures that researchers worldwide can submit their papers with complete confidence that their intellectual property is safe with us.
Easy, secure payment gateways
Pay via credit card
We accept Visa, MasterCard, American Express, Diners Club, and Discover, and have partnered with two payment gateways—PayPal and CC Avenue. The payment gateways available to you will differ depending on your country and the card you choose to use.
Pay via bank transfer
You can make payments directly into our bank account and email the bank wire transfer receipt to us. We recommend bank wire transfer for payments exceeding $1000.
Native English editors - specialists in your subject area guiding you to publication success
Our unique editor-selection algorithm ensures that your paper is assigned to highly qualified language specialists with expertise in your subject. As it learns your requirements, we are able to offer more tailored services. Our editors are all native English speakers who hold PhDs or Master’s degrees from top universities such as Harvard, MIT and Oxford. They are all experts in their respective academic fields with an average of 19.4 years’ experience in editing. Most of them are published authors and peer reviewers in their own right as well as members of some of the world’s best editor societies, such as BELS and CSE.

- Cardiology and Vascular Medicine
- Biomedical Devices and Pediatrics
- Organ Systems (Gastroenterology, Urology, etc.)
- Muscle, Bone, and Physical and Sports Medicine
- Plastic Surgery
- Reproductive Medicine and OBGYN

- Radiotherapy Planning
- Diabetes and Endocrine Research
- Epidemiology

- Clinical Oncology
- Biochemistry and Structural Biology
- Systems Biology and Biostatistics
- Molecular Biology: Embryonic, Cellular, and Evolutionary
- Microbiology and Immunology
Publication Ethics
We at BMJ, and our partners at Editage, are aware of the increasing pressure that the academic research and publishing community are under today. As a leading provider of editorial and publication support services for the past 12 years, we have on occasion been witness to potential cases of ethical violation while editing research papers. A majority of these are instances of “accidental plagiarism” where authors unwittingly engage in unethical practices, mainly because of lack of information and professional training on the subject.
As the first people to look at research papers, even before journal editors do, we can serve as trusted advisors to thousands of researchers, especially early-career scientists not fully aware of the pitfalls of unethical academic publication by:
- Ensuring quality
- Offering processes for suspected ethical misconduct
- Providing educational initiatives for editors and authors
Frequently asked questions
What are the different types of editing services you offer.
We provide manuscript editing services to meet the publication standards of international high-impact journals. We offer 3 levels of manuscript editing and proofreading services for academic research papers.
What training do your editors and writers receive?
All our manuscript editors and writers go through the Basic Editor Training (BET) program that comprises 5 standard modules and various customized programs.
Authors' success stories
“The unique and extremely helpful thing about Editage is its professionalism and punctuality! For non-native authors, Editage makes your writing language more academically and professionally. Thus more chance to getting publish.”
Poowin Bunyavejchewin Walailak University, School of Liberal Arts
It was great to have a native speaker scrutinize our manuscripts. We managed to publish two papers with the help of Editage in good quality international journals. I suggest using Editage to my colleagues to get a flawless manuscript before the publication.
Lukasz Kaczmarek Adam Mickiewicz University
It's really a high quality editing service and I'm quite pleased about the work. If an author is seeking qualified writing and publication support, this is really true address.
Ugur Erdemir Istanbul University, Assc. Prof., Dentistry

UNLOCK YOUR ACADEMIC POTENTIAL WITH HOPE'S SCIENTIFIC WRITING SERVICES
Embark on a journey of academic excellence with Hope's specialized scientific writing services. Our dedicated team is committed to making your academic endeavors smoother and more rewarding. Whether you're facing complex concepts or intricate research, we're here to transform your scientific writing into insightful masterpieces.
Expert Assistance Across Scientific Disciplines
At Hope, we boast a team of seasoned professionals proficient in a wide array of scientific disciplines. From life sciences to food science, statistics to chemistry, our experienced writers are ready to tackle any subject matter with precision and expertise.
Tailored Solutions for Your Convenience
We understand the importance of timely submissions. That's why we offer multiple options for prompt and cost-effective delivery. Whether you need a quick turnaround or prefer a more comprehensive approach, we've got you covered.
Thorough Research, Authentic Results
At Hope, quality assurance is paramount. Our efficient editorial team meticulously proofreads and validates every scientific document to ensure accuracy, coherence, and adherence to academic standards. You can rest assured that your work is in capable hands..
Transform Your Academic Journey with Hope
Let Hope be your trusted companion in navigating the intricate landscape of scientific academia. With our unwavering support and expertise, you can confidently tackle even the most daunting writing tasks and emerge victorious. Contact us today to experience the difference that Hope's scientific writing services can make in your academic pursuits.

Overcome Impossible Deadlines with Instant Assignment Writing Support
Facing daunting deadlines? Seek refuge in our swift assignment writing aid. With an adept team of assignment specialists, we assure flawless submissions even under the tightest schedules. Connect with us and unveil your requirements to kickstart your academic triumph.
Assignment Help For

Why Opt for Hope's Assignment Writing Services?
At Hope, we offer comprehensive solutions tailored to meet all your assignment demands. Yet, our appeal extends beyond mere convenience. Here are six compelling grounds to choose our assignment assistance:
Expertise and Efficiency
Our adept professionals are proficient across various subjects, ensuring meticulous handling of your assignments.
Timely Delivery
We recognize the significance of punctuality, guaranteeing prompt delivery even for urgent tasks.
Customized Solutions
We personalize our services to align with your precise needs, ensuring a bespoke approach to each assignment.
Quality Assurance
Each assignment undergoes stringent quality checks, ensuring adherence to academic standards and excellence in every submission.
Transparent Communication
We prioritize transparent communication, keeping you informed throughout the process for a seamless experience.
Affordable Pricing
Our services are competitively priced, making academic assistance accessible without compromising quality.

Plagiarism-Free Guarantee
At Hope, academic integrity is paramount. We understand the grave consequences of plagiarism and ensure that every assignment is crafted from scratch. Our team conducts thorough research and produces original content, safeguarding your work against any suspicion of plagiarism. With us, you can rest assured that your academic reputation is protected, and your assignments are authentic representations of your knowledge and skills.
Don't succumb to impossible deadlines. Reach out to Hope today and commence your journey towards academic success with confidence.
Ordering your assignment services from Hope is as easy as following four simple steps:
Reach out and share your requirements.
Begin by reaching out to our team for assignment writing services. Share your specific requirements, and we'll carefully review every detail. Once done, we'll send you an invoice detailing the exact price along with instructions on how to make the payment..
Complete Payment and Delegate Your Project
Review the provided price quote and, if you're ready to proceed, complete the payment following the provided instructions. Once the payment is processed, we'll assign your project to our team of professional assignment writers.
Collaborative Development and Feedback
Our writers will dive into meticulous research on your topic, crafting an outline and initial draft of the assignment. We'll then collaborate with you, gathering your feedback on the initial draft. We're committed to ensuring the final version is 100% aligned with your expectations.
Final Polishing and Delivery
In the final stage, we'll initiate our assignment editing services, conducting careful proofreading and validation. Once complete, we'll send the perfectly polished assignment straight to your inbox, ready for submission.
Experience the simplicity and efficiency of Hope's assignment services today, and embark on a journey of academic success with confidence.
Main subjects.
Food Science
Life Science
Pharmaceutical Science

Scientific Writing Services

Essential Data Corporation provides top-notch Scientific Writing Services, but understanding what those services could look like is a key first step.
What is Scientific Writing?
Scientific writing, also known as scientific technical writing, is a specialized form of communication essential in the world of research and academia. This type of writing involves presenting complex scientific information in a clear, concise, and objective manner. Scientific writers play a crucial role in various industries, translating intricate scientific concepts into accessible language for diverse audiences.
What is the Purpose of Scientific Writing Services?
Scientific writing services help researchers share their findings and methodologies, promoting transparency and advancement. They also enable peers to evaluate and validate the research, contributing to evidence-based practices. Additionally, scientific writing services serve as a reference for future studies, encouraging further research and development.
Contact Us for Technical Writing
" * " indicates required fields
What are the Benefits of Scientific Writing Services?
Improved Clarity and Accessibility : Scientific writing services provided by technical writers result in clearer and more accessible scientific content. By simplifying complex information, technical writers make scientific concepts easier to understand for diverse audiences, including researchers, professionals, and the general public.
Ensured Compliance with Scientific Standards : Technical writers ensure that scientific documents adhere to the rigorous style and formatting guidelines required for publication. This adherence to standards enhances the credibility and professionalism of the research, increasing its acceptance by peer-reviewed journals and scientific communities.
Enhanced Organization and Structure : Scientific writing services include effective organization and structuring of scientific content. Technical writers arrange information logically, ensuring that key findings are highlighted and supporting details are presented coherently. This structured approach improves the readability and comprehension of scientific documents.
Customized Audience Engagement : Technical writers tailor scientific content to suit the needs and preferences of specific target audiences. Whether writing for researchers, policymakers, or the general public, they adjust the tone, language, and level of detail to maximize audience engagement and understanding.
Accurate Representation of Research : Scientific writing services provided by technical writers ensure the accurate representation of research findings and data. Technical writers meticulously translate scientific data into written form, ensuring precision, objectivity, and clarity while upholding the integrity of the research.
Efficient Project Management : Hiring a technical writer for scientific writing services streamlines the writing and publication process. Technical writers manage projects efficiently, coordinating timelines, collaborating with researchers and editors, and overseeing the writing process from start to finish. This efficient management ensures timely delivery of high-quality scientific documents.
Increased Publication Success : Scientific writing services offered by technical writers enhance the likelihood of publication acceptance in scientific journals. By optimizing manuscript structure, language, and presentation, technical writers improve the overall quality and appeal of scientific manuscripts, thereby increasing their chances of acceptance by peer reviewers and editors.
Cost and Time Savings : Outsourcing scientific writing tasks to technical writers results in cost and time savings for research organizations. Technical writers expedite the writing process, reduce revision cycles, and minimize resources expended on scientific communication efforts, ultimately delivering cost-effective solutions.

How EDC Can Help with Scientific Writing Services
Whether you need a single technical scientific writer for a brief project or a team of consultants to produce a complete line of documentation, the quality of our work is guaranteed for you. Our clients work closely with an Engagement Manager from one of our 30 local offices for the entire length of your project at no additional cost. Contact us at (800) 221-0093 or [email protected] to get started.
Scientific Writing Related Articles
If you’d like to learn more about scientific writing services and what they can do for you, check out some of our related content below:

Technical Writers: Everything You Need to Know

What to Know About Technical Writing
Technical writing encompasses the art of explaining complex subjects to specific audiences, crucial for businesses, industries, and end-users alike. From user manuals to product plans, technical writers bridge the gap between technical expertise and user comprehension, playing a vital role in modern communication and innovation.

Documentation Writers and Why Most Businesses Need Them
Discover the pivotal role documentation writers play in businesses by ensuring clear, comprehensive technical documentation vital for smooth operations. With expertise in various technical documents, from IT manuals to process documentation, documentation writers are indispensable assets, facilitating effective communication and seamless information flow within organizations.
Contact Us for Scientific Writing Services
Hire EDC technical writers for scientific writing or other technical writing documentation:
